Quantity before Quality: A Topic in Chemistry

When we speak of quantity, what does it mean? I know you’re asking why a Math subject matter is included in Chemistry. LET US FIND OUT!
Speaking of Quantity, we refer to as numbers, statistics, records, ratios and etc. But what is its relation to a topic in chemistry? Do we need to have this confusing statistical analysis and mind-freaking algebraic equations? Well, the answer is NO! All we have to do is to know the quantitative ratios of reactants and products in a chemical equation known as STOICHIOMETRY. Stoichiometry is a big long word which denotes the general process of figuring out how much stuff you need to make something.
In Stoichiometry, we need to have that equal ratios of reactants and products also known to as by the term “balanced chemical equation”. Through the use of this we are able to calculate the amount of products formed in after the chemical reaction maybe in moles, atoms, formula unit, molecules, litre, and grams, the mass of reactants before the reaction, their molecular weight and other factors depending on the question given.
We also have the term Limiting and Excess Reagents. Limiting Reagents are those reactants that limits the amount of products produced in such a way that the reactant is totally consumed in reaction, while the Excess Reagents, well by its root “excess” meaning that some of the amount of the particular substance were not consumed leaving spare amounts of the substance.
Given in some information above, I know you’re mystified by those “other factors” that, indeed, will be needed for some questions. Actually I’m gonna give brief explanations, and introductions to each but first let’s tackle the Avogadro’s number which is needed in some computations.
Amadeo Avogadro an Italian scientist proposed a constant value relative to chemistry. This constant value is 6.0221415 × 1023 derived from the concept stating that the volume of a gas is proportional to the number of atoms or molecules regardless of its nature. The unit of this value varies for elements we have the unit “atoms”, for compounds formed by covalent bonding we have “molecules”, and for those compounds formed by ionic bond we have “formula units.”
Let us first begin with the MASS to MOLE. Given the mass of substance A which is 16.0g, we are opt to find for its equivalent mole. Given that the formula mass is equal to 2.43g. We just have to divide the given amount of the substance to its molar mass. Still wondering? Well, I’m gonna give you its dimensional analysis form.
16.0g H2O * = 0.888 moles of H2O
Let’s move on to another we have the MOLE to MOLE ratio, which is the easiest of them all. For example we have the balanced equation 2H2O 2H2 + O2. We are to find the moles of H2. Let H2O have 0.876moles. First we have to divide the coefficient of H2O,then multiply it to the coefficient of H2. Let’s give it a try shall we?
0.876moles H2O * = 0.876moles of H2
For another part we have the MASS to MASS ratio. For example again we have the balanced equation of 2H2O 2H2 + O2. Given that O2 has a mass of 5.78grams. We have to calculate the mass of the H2O. First we have to divide the given mass of O2 to its molecular mass to find for its moles. Then, we have to use the coefficients of O2 and H2O by dividing the obtained moles of O2 to its coefficient then multiplying it to the coefficient of H2O. So now we have the moles of H2O. Then, the moles of H2O be multiplied to its molar mass. Let us see!
5.78g O2 * * * = 13.0 g H2O
For an extra we also have the MOLE to VOLUME. Again given the balanced equation 2H2O 2H2 + O2. We are to find the volume of O2 if H2 has 1.96moles. First we have to use the coefficients by dividing the given mole of H2O to its coefficient and then multiplying it to the coefficient of O2. Then after getting the moles of O2 we have to multiply it by 22.4L (Note that 22.4L is a constant value resembling the Avogadro’s Number). Let us put this up to its dimensional analysis form.
1.96moles H2 * * = 22.0L O2
In addition we also have the VOLUME to VOLUME. Once again let as assume the never ending balanced chemical equation of 2H2O 2H2 + O2. If we have 26.8L of H2O, how many Litres of O2 do we get? First let us divide the given Litres of H2O to the constant value of 22.4L to have the moles of H2O. Then, using the coefficients we have to divide the moles of H2O to its coefficient and multiply it to the coefficient of O2 and finally multiply it by the constant value of 22.4L. Let us solve it.
26.8L H2O * * * = 13.4L O2
One more part is the MASS to VOLUME. Once again, I think that you already have guessed what’s my example and probably you are right, so we have the never ending balanced chemical equation of 2H2O 2H2 + O2. So if we have 56.9g of H2, what Litres of O2 did we get? First, we have to get the mole of H2 by dividing it by its molecular mass. Then, use the divide the obtained mole to its coefficient then multiply to the coefficient of O2. After that, multiply the mole of O2 to the constant value 22.4L. Let us check it out
56.9g H2 * * * = 315L O2
If you’re incessantly keeping wrong answers, here are some tips regarding stoichiometry. First and foremost you must have the correct formula for compounds, molecules, or elements. Make sure that you also have the correct balanced equation for this may add up for the wrong answers you’re getting. Also look for the correct equation to be used and don’t get messed up by some terms. For example the professor stated Magnesium, but you wrote on your paper Mn, which is wrong because its manganese. Always remember the limiting and excess reagents to know the right amount of products produced. The correct number of significant figures also gives the sunny-side-up of everything. And always remember to recheck all questions for the correction of the mistakes. Hope you all learned something from me! Good Luck young Chemists!
By: Ian Christopher S. Lucas
Speaking of Quantity, we refer to as numbers, statistics, records, ratios and etc. But what is its relation to a topic in chemistry? Do we need to have this confusing statistical analysis and mind-freaking algebraic equations? Well, the answer is NO! All we have to do is to know the quantitative ratios of reactants and products in a chemical equation known as STOICHIOMETRY. Stoichiometry is a big long word which denotes the general process of figuring out how much stuff you need to make something.
In Stoichiometry, we need to have that equal ratios of reactants and products also known to as by the term “balanced chemical equation”. Through the use of this we are able to calculate the amount of products formed in after the chemical reaction maybe in moles, atoms, formula unit, molecules, litre, and grams, the mass of reactants before the reaction, their molecular weight and other factors depending on the question given.
We also have the term Limiting and Excess Reagents. Limiting Reagents are those reactants that limits the amount of products produced in such a way that the reactant is totally consumed in reaction, while the Excess Reagents, well by its root “excess” meaning that some of the amount of the particular substance were not consumed leaving spare amounts of the substance.
Given in some information above, I know you’re mystified by those “other factors” that, indeed, will be needed for some questions. Actually I’m gonna give brief explanations, and introductions to each but first let’s tackle the Avogadro’s number which is needed in some computations.
Amadeo Avogadro an Italian scientist proposed a constant value relative to chemistry. This constant value is 6.0221415 × 1023 derived from the concept stating that the volume of a gas is proportional to the number of atoms or molecules regardless of its nature. The unit of this value varies for elements we have the unit “atoms”, for compounds formed by covalent bonding we have “molecules”, and for those compounds formed by ionic bond we have “formula units.”
Let us first begin with the MASS to MOLE. Given the mass of substance A which is 16.0g, we are opt to find for its equivalent mole. Given that the formula mass is equal to 2.43g. We just have to divide the given amount of the substance to its molar mass. Still wondering? Well, I’m gonna give you its dimensional analysis form.
16.0g H2O * = 0.888 moles of H2O
Let’s move on to another we have the MOLE to MOLE ratio, which is the easiest of them all. For example we have the balanced equation 2H2O 2H2 + O2. We are to find the moles of H2. Let H2O have 0.876moles. First we have to divide the coefficient of H2O,then multiply it to the coefficient of H2. Let’s give it a try shall we?
0.876moles H2O * = 0.876moles of H2
For another part we have the MASS to MASS ratio. For example again we have the balanced equation of 2H2O 2H2 + O2. Given that O2 has a mass of 5.78grams. We have to calculate the mass of the H2O. First we have to divide the given mass of O2 to its molecular mass to find for its moles. Then, we have to use the coefficients of O2 and H2O by dividing the obtained moles of O2 to its coefficient then multiplying it to the coefficient of H2O. So now we have the moles of H2O. Then, the moles of H2O be multiplied to its molar mass. Let us see!
5.78g O2 * * * = 13.0 g H2O
For an extra we also have the MOLE to VOLUME. Again given the balanced equation 2H2O 2H2 + O2. We are to find the volume of O2 if H2 has 1.96moles. First we have to use the coefficients by dividing the given mole of H2O to its coefficient and then multiplying it to the coefficient of O2. Then after getting the moles of O2 we have to multiply it by 22.4L (Note that 22.4L is a constant value resembling the Avogadro’s Number). Let us put this up to its dimensional analysis form.
1.96moles H2 * * = 22.0L O2
In addition we also have the VOLUME to VOLUME. Once again let as assume the never ending balanced chemical equation of 2H2O 2H2 + O2. If we have 26.8L of H2O, how many Litres of O2 do we get? First let us divide the given Litres of H2O to the constant value of 22.4L to have the moles of H2O. Then, using the coefficients we have to divide the moles of H2O to its coefficient and multiply it to the coefficient of O2 and finally multiply it by the constant value of 22.4L. Let us solve it.
26.8L H2O * * * = 13.4L O2
One more part is the MASS to VOLUME. Once again, I think that you already have guessed what’s my example and probably you are right, so we have the never ending balanced chemical equation of 2H2O 2H2 + O2. So if we have 56.9g of H2, what Litres of O2 did we get? First, we have to get the mole of H2 by dividing it by its molecular mass. Then, use the divide the obtained mole to its coefficient then multiply to the coefficient of O2. After that, multiply the mole of O2 to the constant value 22.4L. Let us check it out
56.9g H2 * * * = 315L O2
If you’re incessantly keeping wrong answers, here are some tips regarding stoichiometry. First and foremost you must have the correct formula for compounds, molecules, or elements. Make sure that you also have the correct balanced equation for this may add up for the wrong answers you’re getting. Also look for the correct equation to be used and don’t get messed up by some terms. For example the professor stated Magnesium, but you wrote on your paper Mn, which is wrong because its manganese. Always remember the limiting and excess reagents to know the right amount of products produced. The correct number of significant figures also gives the sunny-side-up of everything. And always remember to recheck all questions for the correction of the mistakes. Hope you all learned something from me! Good Luck young Chemists!
By: Ian Christopher S. Lucas
Stoichiometry

There is a quantitative interpretation of chemical reactions which is called Stoichiometry. It is the relationship of quantities (mass of substance or volume of gas) in a chemical change according to the balanced chemical equation. Chemical Stoichiometry is also the quantitative study among the relationships of the reactants and product. It involves the molar relationships between the products and reactants. The Stoichiometric coefficients in a chemical equation can be interpreted as the number of moles of each substance by the use of mole method.
Here is the general approach for solving stoichiometry problems, first, write a balanced equation for the reaction;
Example: 2 CO (g) + O2 (g) à 2 CO(g)
Second, convert the given amount of the reactant (in grams or other units) to number of moles.
Example: if 9.8 moles of CO react completely with O2 to form CO2, calculate the amount of CO2 produced in moles-moles of CO2 produced= 9.8 mol CO x 2 mol CO2 /2 mol CO=9.8mol CO2
Third step is use the mole ratio from the balanced equation to calculate the number of moles of product formed .
Example: Grams of CO à moles of CO à moles of CO2 à grams of CO2
Lastly, convert the moles of products to grams of products.
Stoichiometry is truly one of the most important topics in Chemistry. It involves the use of the chemical formulas, mole calculations and chemical equations. It is also essential in industry, there, it is used to do cost and analysis for manufacturing chemicals. In fact, there are lots of other uses of Stoichiometric calculations around us.
References: Advanced Chemistry IV
By: Joanna M. Mayo
Here is the general approach for solving stoichiometry problems, first, write a balanced equation for the reaction;
Example: 2 CO (g) + O2 (g) à 2 CO(g)
Second, convert the given amount of the reactant (in grams or other units) to number of moles.
Example: if 9.8 moles of CO react completely with O2 to form CO2, calculate the amount of CO2 produced in moles-moles of CO2 produced= 9.8 mol CO x 2 mol CO2 /2 mol CO=9.8mol CO2
Third step is use the mole ratio from the balanced equation to calculate the number of moles of product formed .
Example: Grams of CO à moles of CO à moles of CO2 à grams of CO2
Lastly, convert the moles of products to grams of products.
Stoichiometry is truly one of the most important topics in Chemistry. It involves the use of the chemical formulas, mole calculations and chemical equations. It is also essential in industry, there, it is used to do cost and analysis for manufacturing chemicals. In fact, there are lots of other uses of Stoichiometric calculations around us.
References: Advanced Chemistry IV
By: Joanna M. Mayo
Mole Method

The mole is a unit of measurement used in chemistry to express amounts of a chemical substance, defined as an amount of a substance that contains as many elementary entities. It is also known as the SI measure of quantity of a “chemical entity”, which can be an atom, molecule, formula unit, electron or photon. It can be classify as Avogadro ’s number, Molar Mass, and Molar Volume. Let’s have a wider view about the given classification.
The first one is the Avogadro’s number, it is the name given to the numerical value of 6.02*10^23 – designated by the Italian physicist, Amedeo Avogadro. There are some things to understand about the Avogadro’s number. One example of this is that, its value can be known only to the precision that the number of atoms in a measurable weight of a substance can be estimated. Here is the illustration of the Avogadro’s number.
1 mole of any element=6.02*10^23 atoms
1 mole of any compound=6.02*10^23 molecules
Here is one example of getting the moles of a certain element. (Take note that you can use either the factor-label method and the ratio and proportion.)
How many moles of magnesium are 3.01*10^22 atoms of magnesium?
moles Mg= 3.01*10^22 atoms Mg x 1 mol Mg
= 5.00*10^-1 mol Mg
The second one is the Molar mass , it is the mass in grams of a substance that is numerically equal to the substance formula mass. While atomic mass is the mass of the atom in atomic mass unit. Also belong is the molecular mass, it is the sum of the atomic masses in the molecule. Here is the illustration.
1 mole of any element= atomic mass
1 mole of any compound= molecular mass
Here is one example of getting the molar quantity of a compound.
How many moles are there in 29.0 grams of NaCl?
NaCl
Na= 1x22.99 = 22.99
Cl= 1x35.45= 35.45 = 58.44 g/mol
? mole NaCl x 1 mole
= .496 mol
The last one is the Molar Volume, means that in 1 mol of any gas in an enclosed container occupies a volume of 22.4 liters at standard temp. and pressure.
Illustration
1 mole of any gas at STP= 22.4 L
Here is one example of getting the molar volume.
What is the volume in L of 16.0 g of oxygen at STP?
16.0 g Oxygen x 1 mol 0xygen x 22.4 L
= 11.2 L
That’s all. I hope you understand well the mole method. :)))
By: Abegail Anne G. Reyes
The first one is the Avogadro’s number, it is the name given to the numerical value of 6.02*10^23 – designated by the Italian physicist, Amedeo Avogadro. There are some things to understand about the Avogadro’s number. One example of this is that, its value can be known only to the precision that the number of atoms in a measurable weight of a substance can be estimated. Here is the illustration of the Avogadro’s number.
1 mole of any element=6.02*10^23 atoms
1 mole of any compound=6.02*10^23 molecules
Here is one example of getting the moles of a certain element. (Take note that you can use either the factor-label method and the ratio and proportion.)
How many moles of magnesium are 3.01*10^22 atoms of magnesium?
moles Mg= 3.01*10^22 atoms Mg x 1 mol Mg
= 5.00*10^-1 mol Mg
The second one is the Molar mass , it is the mass in grams of a substance that is numerically equal to the substance formula mass. While atomic mass is the mass of the atom in atomic mass unit. Also belong is the molecular mass, it is the sum of the atomic masses in the molecule. Here is the illustration.
1 mole of any element= atomic mass
1 mole of any compound= molecular mass
Here is one example of getting the molar quantity of a compound.
How many moles are there in 29.0 grams of NaCl?
NaCl
Na= 1x22.99 = 22.99
Cl= 1x35.45= 35.45 = 58.44 g/mol
? mole NaCl x 1 mole
= .496 mol
The last one is the Molar Volume, means that in 1 mol of any gas in an enclosed container occupies a volume of 22.4 liters at standard temp. and pressure.
Illustration
1 mole of any gas at STP= 22.4 L
Here is one example of getting the molar volume.
What is the volume in L of 16.0 g of oxygen at STP?
16.0 g Oxygen x 1 mol 0xygen x 22.4 L
= 11.2 L
That’s all. I hope you understand well the mole method. :)))
By: Abegail Anne G. Reyes
Mass-Mass Stoichiometry
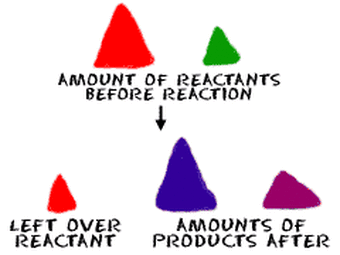
First of all, let me give you a short review in Stoichiometry. Stoichiometry is the calculation of quantities of substances involved in chemical reactions. So mass-mass stoichiometry is so named because the unknown mass of a substance is being calculated from a given mass of a reactant or product in a chemical equation.
Examples:
1. How many grams of Fe are produced if 0.455 g of Ag are used?
· First, balance the given chemical equation.
Fe + 3AgNO3 Fe(NO3)3 + 3Ag
· Second, convert the relevant given mass of substance into moles.
0.455 g Ag X 1 mol Ag = 0.0042 mol Ag
107.87 g Ag
· Third, convert the number of moles of a given mass of a given substance into moles of unknown substance using the coefficients in the balanced chemical equation.
0.0042 mol Ag X 1 mol Fe = 0.0014 mol Fe
3 mol Ag
· Fourth, calculate the mass of the unknown substance by using the molar mass of it.
0.0014 mol Fe X 55.85 g Fe = 0.078 g Fe
1 mol Fe
That’s how we calculate the mass of the unknown substance from the given mass of the reactant or product.
Here is another example for you to understand more the mass-mass stoichiometry.
2. How many grams of potassium chloride are produced if 15g of potassium chlorate decompose?
2KClO3 2KCl + 3O2
Solution:
15 g KClO3 X 1 mol KClO3 X 2 mol KCl X 74.55 g KCl = 9.1 g KCl
122.55 g KClO3 2 mol KClO3 1 mol KCl
Or you can calculate this by using the ratio and proportion method.
15 g KClO3 = X g KCl = 9.1 g KCl
2(122.55 g) KClO3 2(74.55 g) KCl
By: Rhea Ciarina B. Ramos
Examples:
1. How many grams of Fe are produced if 0.455 g of Ag are used?
· First, balance the given chemical equation.
Fe + 3AgNO3 Fe(NO3)3 + 3Ag
· Second, convert the relevant given mass of substance into moles.
0.455 g Ag X 1 mol Ag = 0.0042 mol Ag
107.87 g Ag
· Third, convert the number of moles of a given mass of a given substance into moles of unknown substance using the coefficients in the balanced chemical equation.
0.0042 mol Ag X 1 mol Fe = 0.0014 mol Fe
3 mol Ag
· Fourth, calculate the mass of the unknown substance by using the molar mass of it.
0.0014 mol Fe X 55.85 g Fe = 0.078 g Fe
1 mol Fe
That’s how we calculate the mass of the unknown substance from the given mass of the reactant or product.
Here is another example for you to understand more the mass-mass stoichiometry.
2. How many grams of potassium chloride are produced if 15g of potassium chlorate decompose?
2KClO3 2KCl + 3O2
Solution:
15 g KClO3 X 1 mol KClO3 X 2 mol KCl X 74.55 g KCl = 9.1 g KCl
122.55 g KClO3 2 mol KClO3 1 mol KCl
Or you can calculate this by using the ratio and proportion method.
15 g KClO3 = X g KCl = 9.1 g KCl
2(122.55 g) KClO3 2(74.55 g) KCl
By: Rhea Ciarina B. Ramos
STOICHIOMETRY: MASS-MASS PROBLEMS

Stoichiometry deals with the quantities of the substances that enter into, and are produced by chemical reactions. It provides the quantitative relationship between reactants and products in a certain chemical reaction. When we are asked to find out the mass of the unknown given substance, it is called MASS-MASS STOICHIOMETRY. In this problem, we are going to calculate the mass of a substance from a given mass of a reactant or product in a chemical equation.
In this problem, we are going to consider the following:
Ø We need to balance the chemical equation properly.
Ø Convert the given mass of a substance into moles by using the molar mass.
Ø Convert the number of moles of a given mass into moles of unknown substance using the coefficients from the balanced equation.
Ø Calculate the mass of the unknown substance by using the molar mass of the unknown as a unit factor.
Example:
How many grams of chlorine can we obtained from the composition of 64.0 g of AuCl3 by this reaction: 2 AuCl3 → 2 Au + 3 Cl2. Given the balanced equation,
a.) Calculate the moles of AuCl3 when the molar mass of AuCl3= 303.32 g/mol
64.0 g AuCl3 X 1 mol AuCl3 = 0.211 mol AuCl3
303.32 g AuCl3
b.) Convert the number of moles to moles of Cl2 by applying the coefficient from the balanced chemical equation
0.211 mol AuCl3 x 3 mol Cl2 = 0.317 mol Cl2
2 mol AuCl3
c.) Use the mol of Cl2 as a unit factor to obtain the mass of Cl2 that reacts with 64.0 g of AuCl3
0.317 mol Cl2 x 70.9 g Cl2 = 23.0 g Cl2
1 mol Cl2
Or you can have:
64.0 g AuCl3 X 1 mol AuCl3 x 3 mol Cl2 x 70.9 g Cl2 = 23.0 g Cl2
303.32 g AuCl3 2 mol AuCl3 1 mol Cl2
TAKE NOTE!!!
Make sure that the given chemical equation is correctly balanced, if not then balanced it properly.
REFERENCES:
Lectures/Notes in Advance Chemistry
Advance Study Assignment
http://www.chemistry.co.nz/stoichiometry.htm
http://www.chemteam.info/Stoichiometry/Mass-Mass.html
By: Dannah Lyn Antonio
In this problem, we are going to consider the following:
Ø We need to balance the chemical equation properly.
Ø Convert the given mass of a substance into moles by using the molar mass.
Ø Convert the number of moles of a given mass into moles of unknown substance using the coefficients from the balanced equation.
Ø Calculate the mass of the unknown substance by using the molar mass of the unknown as a unit factor.
Example:
How many grams of chlorine can we obtained from the composition of 64.0 g of AuCl3 by this reaction: 2 AuCl3 → 2 Au + 3 Cl2. Given the balanced equation,
a.) Calculate the moles of AuCl3 when the molar mass of AuCl3= 303.32 g/mol
64.0 g AuCl3 X 1 mol AuCl3 = 0.211 mol AuCl3
303.32 g AuCl3
b.) Convert the number of moles to moles of Cl2 by applying the coefficient from the balanced chemical equation
0.211 mol AuCl3 x 3 mol Cl2 = 0.317 mol Cl2
2 mol AuCl3
c.) Use the mol of Cl2 as a unit factor to obtain the mass of Cl2 that reacts with 64.0 g of AuCl3
0.317 mol Cl2 x 70.9 g Cl2 = 23.0 g Cl2
1 mol Cl2
Or you can have:
64.0 g AuCl3 X 1 mol AuCl3 x 3 mol Cl2 x 70.9 g Cl2 = 23.0 g Cl2
303.32 g AuCl3 2 mol AuCl3 1 mol Cl2
TAKE NOTE!!!
Make sure that the given chemical equation is correctly balanced, if not then balanced it properly.
REFERENCES:
Lectures/Notes in Advance Chemistry
Advance Study Assignment
http://www.chemistry.co.nz/stoichiometry.htm
http://www.chemteam.info/Stoichiometry/Mass-Mass.html
By: Dannah Lyn Antonio
MASS TO MASS STOICHIOMETRY

I have learn that Stoichiometry is a prediction of how much of one substance will react or be produced in a chemical reaction relative to the amount of another substance in the reaction.
Mole to mole stoichiometry is very important component in chemistry. It will allow us to convert from one substance to another within the confines of a chemical reaction.
Mass to Mass stoichiometry requires three steps to complete. A diagram is below to make this process more clear:
So, let’s imagine that we are performing this reaction:
H2CO3 + 2 KOH --- K2CO3 + 2 H2O
Using the above equation, suppose we wanted to know how many grams of water (H2O) can be made from 40 g of H2CO3. We know how to convert moles of H2CO3 into moles of H2O, so we need to convert the 40 grams of H2CO3 into moles of H2CO3.
The work looks like this:
Then, we can convert moles of H2CO3 into moles of H2O:
Finally, we do not want our answer in moles, but in grams of H2O, so we complete one more conversion:
Reference:
http://ths.talawanda.net/~BrambleN/classroom/Chemistry/Notes/Section%202C/MassStoichiometry.htm
By: Lori Anne Anda
Mole to mole stoichiometry is very important component in chemistry. It will allow us to convert from one substance to another within the confines of a chemical reaction.
Mass to Mass stoichiometry requires three steps to complete. A diagram is below to make this process more clear:
So, let’s imagine that we are performing this reaction:
H2CO3 + 2 KOH --- K2CO3 + 2 H2O
Using the above equation, suppose we wanted to know how many grams of water (H2O) can be made from 40 g of H2CO3. We know how to convert moles of H2CO3 into moles of H2O, so we need to convert the 40 grams of H2CO3 into moles of H2CO3.
The work looks like this:
Then, we can convert moles of H2CO3 into moles of H2O:
Finally, we do not want our answer in moles, but in grams of H2O, so we complete one more conversion:
Reference:
http://ths.talawanda.net/~BrambleN/classroom/Chemistry/Notes/Section%202C/MassStoichiometry.htm
By: Lori Anne Anda

