Let us Tangle with the Angles
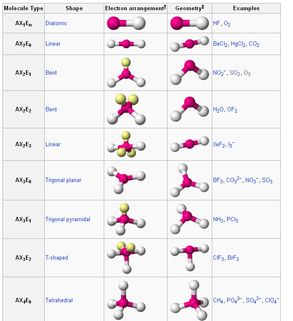
In chemistry, the polarity, reactivity, phase of matter, color, magentism, as well as the biological activity may be determined with angles. How? Well, it’s because of the angles in their molecular structure or what we may call Molecular Geometry.
To determine the shapes of molecules, we must become familiar with the Lewis electron dot structure. Although the Lewis theory does not determine the shapes of molecules, it is the first step in predicting shapes of molecules. The Lewis structure helps us identify the bond pairs and the lone pairs. Then, with the Lewis structure, we apply the valence-shell electron-pair repulsion (VSPER) theory to determine the molecular geometry and the electron-group geometry. To identify and have a complete description of the three-dimensional shape of a molecule, we need to know also learn about the state of bond angle as well.
What is VSPER? The valence-shell electron-pair repulsion (VSEPR) theory states that electron pairs repel each other whether or not they are in bond pairs or in lone pairs. Thus, electron pairs will spread themselves as far from each other as possible to minimize repulsion. VSEPR focuses not only on electron pairs, but it also focus on electron groups as a whole. An electron group can be an electron pair, a lone pair, a single unpaired electron, a double bond or a triple bond on the center atom. Using the VSEPR theory, the electron bond pairs and lone pairs on the center atom will help us predict the shape of a molecule.
The shape of a molecule is determined by the location of the nuclei and its electrons. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. Although VSEPR theory predicts the distribution of the electrons, we have to take in consideration of the actual determinant of the molecular shape.
We must take note that molecular geometry depends not only on the number of electron groups, but also on the number of lone pairs. When the electron groups are all bond pairs, they are named exactly like the electron-group geometry.
We must also take into relation the VSEPR notation. The VSEPR notation is represented by “A” and “X”. The letter “A” represents the central atom and “X” represents the element/compound bonded with the central atom. When lone pairs are present, the letter “E” is added. Subscript represented as “n” are the number of elements/ compounds bonded with the central atom or the number of lone pairs.
We have different electron pair geometry and we are going to discuss them one by one. We have the linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
In linear (AX2), the total numbers of group of electrons is two. Having an approximate bonding angle of 180º. Number of bonding direction is two. Shape is linear. Examples are BeH2 and CO2.
In trigonal planar we have two. The trigonal planar (AX3) having the total numbers of group of electrons is three. Having an approximate bonding angle of 120º. Number of bonding direction is three. Examples are BF3 and NO3. The second is the Bent (AX2E). Having an approximate bonding angle of 120º. Number of bonding direction is two. Example is SO2.
In tetrahedral we have thee. The Tetrahedral (AX4) having the total numbers of group of electrons is four. Having an approximate bonding angle of 109.5º. Number of bonding direction is four. Example is CH4. The second is the Trigonal Pyramidal (AX3E). Number of bonding direction is three. Example is NH3. The third is the Bent (AX2E2). Number of bonding direction is two. Example is H2O.
In trigonal bipyramidal we have four. Having bonding angles of 120º. in plane and 90º in above and below. The trigonal bipyramidal (AX5) having the total numbers of group of electrons is five. Number of bonding direction is five. Example is PCl5. The second is the seesaw (AX4E). Number of bonding direction is four. Example is SF4. The third is the T-shaped (AX3E2). Number of bonding direction is three. Example is ClF3. The fourth is the Linear (AX2E3). Number of bonding direction is two. Example is XeF2.
In octahedral we have five. Having bonding angles of 90º. The Octahedral having the total numbers of group of electrons is six. Number of bonding direction is six. Example is SF6. The second is the square pyramidal (AX5E). Number of bonding direction is five. Example is BrF5. The third is the square planar (AX4E2). Number of bonding direction is four. Example is XeF4. The fourth is the T-shaped (AX3E3). Number of bonding direction is three. The fifth is the linear (AX2E4). Number of bonding direction is two.
By: Ian Christopher S. Lucas
To determine the shapes of molecules, we must become familiar with the Lewis electron dot structure. Although the Lewis theory does not determine the shapes of molecules, it is the first step in predicting shapes of molecules. The Lewis structure helps us identify the bond pairs and the lone pairs. Then, with the Lewis structure, we apply the valence-shell electron-pair repulsion (VSPER) theory to determine the molecular geometry and the electron-group geometry. To identify and have a complete description of the three-dimensional shape of a molecule, we need to know also learn about the state of bond angle as well.
What is VSPER? The valence-shell electron-pair repulsion (VSEPR) theory states that electron pairs repel each other whether or not they are in bond pairs or in lone pairs. Thus, electron pairs will spread themselves as far from each other as possible to minimize repulsion. VSEPR focuses not only on electron pairs, but it also focus on electron groups as a whole. An electron group can be an electron pair, a lone pair, a single unpaired electron, a double bond or a triple bond on the center atom. Using the VSEPR theory, the electron bond pairs and lone pairs on the center atom will help us predict the shape of a molecule.
The shape of a molecule is determined by the location of the nuclei and its electrons. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. Although VSEPR theory predicts the distribution of the electrons, we have to take in consideration of the actual determinant of the molecular shape.
We must take note that molecular geometry depends not only on the number of electron groups, but also on the number of lone pairs. When the electron groups are all bond pairs, they are named exactly like the electron-group geometry.
We must also take into relation the VSEPR notation. The VSEPR notation is represented by “A” and “X”. The letter “A” represents the central atom and “X” represents the element/compound bonded with the central atom. When lone pairs are present, the letter “E” is added. Subscript represented as “n” are the number of elements/ compounds bonded with the central atom or the number of lone pairs.
We have different electron pair geometry and we are going to discuss them one by one. We have the linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
In linear (AX2), the total numbers of group of electrons is two. Having an approximate bonding angle of 180º. Number of bonding direction is two. Shape is linear. Examples are BeH2 and CO2.
In trigonal planar we have two. The trigonal planar (AX3) having the total numbers of group of electrons is three. Having an approximate bonding angle of 120º. Number of bonding direction is three. Examples are BF3 and NO3. The second is the Bent (AX2E). Having an approximate bonding angle of 120º. Number of bonding direction is two. Example is SO2.
In tetrahedral we have thee. The Tetrahedral (AX4) having the total numbers of group of electrons is four. Having an approximate bonding angle of 109.5º. Number of bonding direction is four. Example is CH4. The second is the Trigonal Pyramidal (AX3E). Number of bonding direction is three. Example is NH3. The third is the Bent (AX2E2). Number of bonding direction is two. Example is H2O.
In trigonal bipyramidal we have four. Having bonding angles of 120º. in plane and 90º in above and below. The trigonal bipyramidal (AX5) having the total numbers of group of electrons is five. Number of bonding direction is five. Example is PCl5. The second is the seesaw (AX4E). Number of bonding direction is four. Example is SF4. The third is the T-shaped (AX3E2). Number of bonding direction is three. Example is ClF3. The fourth is the Linear (AX2E3). Number of bonding direction is two. Example is XeF2.
In octahedral we have five. Having bonding angles of 90º. The Octahedral having the total numbers of group of electrons is six. Number of bonding direction is six. Example is SF6. The second is the square pyramidal (AX5E). Number of bonding direction is five. Example is BrF5. The third is the square planar (AX4E2). Number of bonding direction is four. Example is XeF4. The fourth is the T-shaped (AX3E3). Number of bonding direction is three. The fifth is the linear (AX2E4). Number of bonding direction is two.
By: Ian Christopher S. Lucas
Seek to Chemistry (Crossword)
H A I L G E O M E T R Y F U N M O L E C U L A R G E O M E T R Y R
E E C P O P L E A S E D O N T L E T M E G O H O M O L Y T I C I A
T Q H A B N B L U N T A R R H E N I U S A C I D L I A R S T H S A
E U E I O U E L I N E A R S U L T S U C K E R A C I D I C E O T M
R I M N R N A P T H Y A M P H I P R O T I C O N U S C S A M L R P
O L I T C N X Z A S O R R Y K U Y A A R U S X G T T I O R P D A H
G I C O I I W K L I C E L E S T I A L H S E Y E T A S M R E O N I
E B A Y X E U B J I R R E V E R S I B L E L G L I N S E E R N G P
N R L C O N J U G A T E B A S E B O R O N D E S N D T R D A X E R
E I S P T I N F I N I T Y S I G R P R A Y E R S G L I S P T V R O
O U Z Q K L Z B R E A K W A T E R L E W I S B A S E L D R U I S B
U M F H Y B R I D I Z A T I O N H Y D R O G E N L G L I A R C T Y
S H O N E E Q U I L I B R I U M C O N S T A N T I T B A S E J O E
E E C P O P L E A S E D O N T L E T M E G O H O M O L Y T I C I A
T Q H A B N B L U N T A R R H E N I U S A C I D L I A R S T H S A
E U E I O U E L I N E A R S U L T S U C K E R A C I D I C E O T M
R I M N R N A P T H Y A M P H I P R O T I C O N U S C S A M L R P
O L I T C N X Z A S O R R Y K U Y A A R U S X G T T I O R P D A H
G I C O I I W K L I C E L E S T I A L H S E Y E T A S M R E O N I
E B A Y X E U B J I R R E V E R S I B L E L G L I N S E E R N G P
N R L C O N J U G A T E B A S E B O R O N D E S N D T R D A X E R
E I S P T I N F I N I T Y S I G R P R A Y E R S G L I S P T V R O
O U Z Q K L Z B R E A K W A T E R L E W I S B A S E L D R U I S B
U M F H Y B R I D I Z A T I O N H Y D R O G E N L G L I A R C T Y
S H O N E E Q U I L I B R I U M C O N S T A N T I T B A S E J O E
1. Refers to the arrangement of the atoms within a given molecule
2. State where no observable changes as time goes by
3. It is a type of molecular structure that has four bonds all on one central atom, with no extra unshared electron pairs
4. Substance that acts either as an acid or base
5. The mixing of native orbitals to form a newly made hybrid orbitals
6. A substance that dissociates in water to form hydrogen ions
7. A valence electron pair without bonding or sharing with other atoms
8. Term used when a product formed from the reaction may not be combined to form the reactants
9. A compound that can donate an electron pair
10. Types of molecules that share a chemical formula but have different geometries resulting in very different properties
11. Chemical bond dissociation of a neutral molecule generating two free radicals
12. Specimen remaining after the removal of one proton
13. Reactions which have different phases
14. A condition needed to convert products back to its reactants
15. The ratio of concentrations when equilibrium is reached in a reversible reaction
By: Brea Jaye N. Uy
2. State where no observable changes as time goes by
3. It is a type of molecular structure that has four bonds all on one central atom, with no extra unshared electron pairs
4. Substance that acts either as an acid or base
5. The mixing of native orbitals to form a newly made hybrid orbitals
6. A substance that dissociates in water to form hydrogen ions
7. A valence electron pair without bonding or sharing with other atoms
8. Term used when a product formed from the reaction may not be combined to form the reactants
9. A compound that can donate an electron pair
10. Types of molecules that share a chemical formula but have different geometries resulting in very different properties
11. Chemical bond dissociation of a neutral molecule generating two free radicals
12. Specimen remaining after the removal of one proton
13. Reactions which have different phases
14. A condition needed to convert products back to its reactants
15. The ratio of concentrations when equilibrium is reached in a reversible reaction
By: Brea Jaye N. Uy
Acids and Bases

For thousands of years, we all know that vinegar, lemon juice, oranges and many other foods taste sour. However, some of us don’t have any idea why these things taste sour. The fact is because these things are all acids. We also hear bases all the time. But do we even know what bases are? In this lesson, you will learn about these things we called acids and bases.
Acids taste sour, are corrosive to metals, change litmus (a dye extracted from lichens) red, and become less acidic when mixed with bases.
Bases feel slippery, change litmus blue, and become less basic when mixed with acids.
DEFINITION OF ACIDS AND BASES
· Arrhenius Definition
In the late 1800s, the Swedish scientist Svante Arrhenius proposed that water can dissolve many compounds by separating them into their individual ions. Arrhenius suggested that acids are compounds that contain hydrogen and can dissolve in water to release hydrogen ions into solution. For example, hydrochloric acid (HCl) dissolves in water as follows:
HCl -----------> H+(aq) + Cl-(aq)
Arrhenius defined bases as substances that dissolve in water to release hydroxide ions (OH-) into solution. For example, a typical base according to the Arrhenius definition is sodium hydroxide (NaOH):
NaOH ---------------> Na+(aq) + OH-(aq)
· Bronsted-Lowry Definition
In 1923, the Danish scientist Johannes Brønsted and the Englishman Thomas Lowry published independent yet similar papers that refined Arrhenius' theory. In Brønsted's words, "... acids and bases are substances that are capable of splitting off or taking up hydrogen ions, respectively." The Brønsted-Lowry definition broadened the Arrhenius concept of acids and bases.
According to Brønsted–Lowry definition, an acid is a compound that can donate a proton, and a base is a compound that can receive a proton. An acid–base reaction is, thus, the removal of a hydrogen ion from the acid and its addition to the base. This does not refer to the removal of a proton from the nucleus of an atom, which would require levels of energy not attainable through the simple dissociation of acids, but to removal of a hydrogen ion (H+).
The removal of a proton (hydrogen ion) from an acid produces its conjugate base, which is the acid with a hydrogen ion removed, and the reception of a proton by a base produces its conjugate acid, which is the base with a hydrogen ion added.
For example, the removal of H+ from hydrochloric acid (HCl) produces the chloride ion (Cl−), the conjugate base of the acid:
HCl ----------> H+(aq) + Cl-(aq)
The addition of H+ to the hydroxide ion (OH−), a base, produces water (H2O), its conjugate acid:
H+(aq) + OH− ---------------> H2O
· Lewis Definition
In 1923, Gilbert N. Lewis suggested another way of looking at the reaction between H+ and OH- ions. Instead of defining acid–base reactions in terms of protons or other bonded substances, the Lewis definition defines a base (referred to as a Lewis base) to be a compound that can donate an electron pair, and an acid (a Lewis acid) to be a compound that can receive this electron pair.
A Lewis acid is therefore any substance, such as the H+ ion, that can accept a pair of nonbonding electrons. In other words, a Lewis acid is an electron-pair acceptor. A Lewis base is any substance, such as the OH- ion, that can donate a pair of nonbonding electrons. A Lewis base is therefore an electron-pair donor.
HCl + NaOH --------------------> H2O + NaCl
The Lewis definition does not regard this reaction as the formation of salt and water or the transfer of H+ from HCl to OH−. Instead, it regards the acid to be the H+ ion itself, and the base to be the OH− ion, which has an unshared electron pair. Therefore, the acid–base reaction here, according to the Lewis definition, is the donation of the electron pair from OH−to the H+ ion. This forms a covalent bond between H+ and OH−, thus producing water (H2O).
By: Rhea Ciarina B. Ramos
Acids taste sour, are corrosive to metals, change litmus (a dye extracted from lichens) red, and become less acidic when mixed with bases.
Bases feel slippery, change litmus blue, and become less basic when mixed with acids.
DEFINITION OF ACIDS AND BASES
· Arrhenius Definition
In the late 1800s, the Swedish scientist Svante Arrhenius proposed that water can dissolve many compounds by separating them into their individual ions. Arrhenius suggested that acids are compounds that contain hydrogen and can dissolve in water to release hydrogen ions into solution. For example, hydrochloric acid (HCl) dissolves in water as follows:
HCl -----------> H+(aq) + Cl-(aq)
Arrhenius defined bases as substances that dissolve in water to release hydroxide ions (OH-) into solution. For example, a typical base according to the Arrhenius definition is sodium hydroxide (NaOH):
NaOH ---------------> Na+(aq) + OH-(aq)
· Bronsted-Lowry Definition
In 1923, the Danish scientist Johannes Brønsted and the Englishman Thomas Lowry published independent yet similar papers that refined Arrhenius' theory. In Brønsted's words, "... acids and bases are substances that are capable of splitting off or taking up hydrogen ions, respectively." The Brønsted-Lowry definition broadened the Arrhenius concept of acids and bases.
According to Brønsted–Lowry definition, an acid is a compound that can donate a proton, and a base is a compound that can receive a proton. An acid–base reaction is, thus, the removal of a hydrogen ion from the acid and its addition to the base. This does not refer to the removal of a proton from the nucleus of an atom, which would require levels of energy not attainable through the simple dissociation of acids, but to removal of a hydrogen ion (H+).
The removal of a proton (hydrogen ion) from an acid produces its conjugate base, which is the acid with a hydrogen ion removed, and the reception of a proton by a base produces its conjugate acid, which is the base with a hydrogen ion added.
For example, the removal of H+ from hydrochloric acid (HCl) produces the chloride ion (Cl−), the conjugate base of the acid:
HCl ----------> H+(aq) + Cl-(aq)
The addition of H+ to the hydroxide ion (OH−), a base, produces water (H2O), its conjugate acid:
H+(aq) + OH− ---------------> H2O
· Lewis Definition
In 1923, Gilbert N. Lewis suggested another way of looking at the reaction between H+ and OH- ions. Instead of defining acid–base reactions in terms of protons or other bonded substances, the Lewis definition defines a base (referred to as a Lewis base) to be a compound that can donate an electron pair, and an acid (a Lewis acid) to be a compound that can receive this electron pair.
A Lewis acid is therefore any substance, such as the H+ ion, that can accept a pair of nonbonding electrons. In other words, a Lewis acid is an electron-pair acceptor. A Lewis base is any substance, such as the OH- ion, that can donate a pair of nonbonding electrons. A Lewis base is therefore an electron-pair donor.
HCl + NaOH --------------------> H2O + NaCl
The Lewis definition does not regard this reaction as the formation of salt and water or the transfer of H+ from HCl to OH−. Instead, it regards the acid to be the H+ ion itself, and the base to be the OH− ion, which has an unshared electron pair. Therefore, the acid–base reaction here, according to the Lewis definition, is the donation of the electron pair from OH−to the H+ ion. This forms a covalent bond between H+ and OH−, thus producing water (H2O).
By: Rhea Ciarina B. Ramos
Rule Breakers
Have you ever wondered why Chemistry is so complicated yet interesting, especially when you can understand its mind-blowing concepts? When you can answer its questions like why do atoms bond together? Even though we know that two hydrogen atoms combine with one oxygen atom to form water, but why would hydrogen and oxygen "want" to bond with each other?
These questions were interrelated to one another and we can easily answer them through Chemistry’s Octet Rule. We are familiar that atoms have a certain number of electrons in their outer shell called valance electron. These are the electrons that are important when considering whether an atom will bond with one another, as well as the ratio of elements in the compound. So when considering valence electrons, the magic number happens to be eight that’s why it’s called “Octet” rule. When there are eight electrons in the outer or last shell of an atom, it is considered to be "full" and optimal. The reason atoms "want" a full outer shell is because a full outer shell will create a more stable particle. Atoms are always reacting with others to become stable, meaning they want to have a full outer shell.
But as always, rules are made to be broken according to an old saying. In this case, the octet rule has more elements breaking the rule than following it. This is a list of the three classes of exceptions to the octet rule.
First, molecules with an odd number of electrons like nitrous oxide (NO). We can verify this using the Lewis structure. Its bonding structure was illustrated below:
N O
a. Total electrons: 6+5=11
b. Octet on outer elements:
N O
c. Remainder of electrons (11-8=3) on “central” atom:
N O
d. There are currently 5 valence electrons around the nitrogen. A double bond would place 7 around the nitrogen, and triple bond would place 9 around the nitrogen.
The second exception is when molecules in which an atom has less than an octet. This is most often encountered with elements of Boron and Beryllium. Use the Lewis structure for Boron trifluoride (BF3).
F
a. Add electrons (3*7)+3=24
b. Draw connectivities (as illustrated on the left)
B
F F
c. Add octets to outer atoms:
(as illustrated on the right)
d. Add extra electrons (24-24=0) to central atom:
(as illustrated on the right)
e. Does central electron have octet?
* No, it has 6 electrons
* Add a multiple bond (double bond) to see if central atom can achieve
an octet: (as illustrated below)
f. The central Boron now has an octet (there would be three resonance Lewis structures)
The last exception is when molecules in which an atom has more than an octet. This exception can be encounter to ICl4.
a. Count up the valence electrons: 7+(4*7)+1=36 electrons
b. Draw the connectivities:
Cl
Cl I Cl
Cl
c. Add octet of electrons to outer atoms:
d. Add extra electrons (36-32=4) to central atom:
e. The ICl4- Ion thus has 12 valence electrons around the central Iodine
(in the 5d orbitals)
And that’s how easy the Octet rule. Hope it will really help you a lot for you to enjoy Chemistry more! Just focus, analyze and have fun.
By: Joanna M. Mayo
These questions were interrelated to one another and we can easily answer them through Chemistry’s Octet Rule. We are familiar that atoms have a certain number of electrons in their outer shell called valance electron. These are the electrons that are important when considering whether an atom will bond with one another, as well as the ratio of elements in the compound. So when considering valence electrons, the magic number happens to be eight that’s why it’s called “Octet” rule. When there are eight electrons in the outer or last shell of an atom, it is considered to be "full" and optimal. The reason atoms "want" a full outer shell is because a full outer shell will create a more stable particle. Atoms are always reacting with others to become stable, meaning they want to have a full outer shell.
But as always, rules are made to be broken according to an old saying. In this case, the octet rule has more elements breaking the rule than following it. This is a list of the three classes of exceptions to the octet rule.
First, molecules with an odd number of electrons like nitrous oxide (NO). We can verify this using the Lewis structure. Its bonding structure was illustrated below:
N O
a. Total electrons: 6+5=11
b. Octet on outer elements:
N O
c. Remainder of electrons (11-8=3) on “central” atom:
N O
d. There are currently 5 valence electrons around the nitrogen. A double bond would place 7 around the nitrogen, and triple bond would place 9 around the nitrogen.
The second exception is when molecules in which an atom has less than an octet. This is most often encountered with elements of Boron and Beryllium. Use the Lewis structure for Boron trifluoride (BF3).
F
a. Add electrons (3*7)+3=24
b. Draw connectivities (as illustrated on the left)
B
F F
c. Add octets to outer atoms:
(as illustrated on the right)
d. Add extra electrons (24-24=0) to central atom:
(as illustrated on the right)
e. Does central electron have octet?
* No, it has 6 electrons
* Add a multiple bond (double bond) to see if central atom can achieve
an octet: (as illustrated below)
f. The central Boron now has an octet (there would be three resonance Lewis structures)
The last exception is when molecules in which an atom has more than an octet. This exception can be encounter to ICl4.
a. Count up the valence electrons: 7+(4*7)+1=36 electrons
b. Draw the connectivities:
Cl
Cl I Cl
Cl
c. Add octet of electrons to outer atoms:
d. Add extra electrons (36-32=4) to central atom:
e. The ICl4- Ion thus has 12 valence electrons around the central Iodine
(in the 5d orbitals)
And that’s how easy the Octet rule. Hope it will really help you a lot for you to enjoy Chemistry more! Just focus, analyze and have fun.
By: Joanna M. Mayo
Acid-Base Indicators
ACID-BASE INDICATORS
Having trouble with acids and bases? You can’t determine if what category do they belong? Well, some of your questions can answer by this article.
What is an acid-base indicator? An acid-base indicator is a weak acid or a weak base. The undissociated form of the indicator is a different color than the iogenic form of the indicator. An Indicator does not change color from pure acid to pure alkaline at specific hydrogen ion concentration, but rather, color change occurs over a range of hydrogen ion concentrations. This range is termed the color change interval. It is expressed as a pH range.
How is an indicator used? Weak acids are titrated in the presence of indicators which change under slightly alkaline conditions. Weak bases should be titrated in the presence of indicators which change under slightly acidic conditions.
What are some common acid-base indicators? Several acid-base indicators are listed below, some more than once if they can be used over multiple pH ranges. Quantity of indicator in aqueous (aq.) or alcohol (alc.) solution is specified. Tried-and-true indicators include: thymol blue, tropeolin OO, methyl yellow, methyl orange, bromphenol blue, bromcresol green, methyl red, bromthymol blue, phenol red, neutral red, phenolphthalein, thymolphthalein, alizarin yellow, tropeolin O, nitramine, and trinitrobenzoic acid. Data in this table are for sodium salts of thymol blue, bromphenol blue, tetrabromphenol blue, bromcresol green, methyl red, bromthymol blue, phenol red, and cresol red.
Table of Common Acid-Base Indicators
Indicator pH Range Quantity per 10 ml Acid Base
Thymol Blue 1.2-2.8 1-2 drops 0.1% soln. in aq. red yellow
Pentamethoxy red 1.2-2.3 1 drop 0.1% soln. in 70% alc. red-violet colorless
Tropeolin OO 1.3-3.2 1 drop 1% aq. soln. red yellow
2,4-Dinitrophenol 2.4-4.0 1-2 drops 0.1% soln. in 50% alc. colorless yellow
Methyl yellow 2.9-4.0 1 drop 0.1% soln. in 90% alc. red yellow
Methyl orange 3.1-4.4 1 drop 0.1% aq. soln. red orange
Bromphenol blue 3.0-4.6 1 drop 0.1% aq. soln. yellow blue-violet
Tetrabromphenol blue 3.0-4.6 1 drop 0.1% aq. soln. yellow blue
Alizarin sodium
sulfonate 3.7-5.2 1 drop 0.1% aq. soln. yellow violet
α-Naphthyl red 3.7-5.0 1 drop 0.1% soln. in 70% alc. red yellow
p-Ethoxychrysoidine 3.5-5.5 1 drop 0.1% aq. soln. red yellow
Bromcresol green 4.0-5.6 1 drop 0.1% aq. soln. yellow blue
Methyl red 4.4-6.2 1 drop 0.1% aq. soln. red yellow
Bromcresol purple 5.2-6.8 1 drop 0.1% aq. soln. yellow purple
Chlorphenol red 5.4-6.8 1 drop 0.1% aq. soln. yellow red
Bromphenol blue 6.2-7.6 1 drop 0.1% aq. soln. yellow blue
p-Nitrophenol 5.0-7.0 1-5 drops 0.1% aq. soln. colorless yellow
Azolitmin 5.0-8.0 5 drops 0.5% aq. soln. red blue
Phenol red 6.4-8.0 1 drop 0.1% aq. soln. yellow red
Neutral red 6.8-8.0 1 drop 0.1% soln. in 70% alc. red yellow
Rosolic acid 6.8-8.0 1 drop 0.1% soln. in 90% alc. yellow red
Cresol red 7.2-8.8 1 drop 0.1% aq. soln. yellow red
α-Naphtholphthalein 7.3-8.7 1-5 drops 0.1% soln. in 70% alc. rose green
Tropeolin OOO 7.6-8.9 1 drop 0.1% aq. soln. yellow rose-red
Thymol blue 8.0-9.6 1-5 drops 0.1% aq. soln. yellow blue
Phenolphthalein 8.0-10.0 1-5 drops 0.1% soln. in 70% alc. colorless red
α-Naphtholbenzein 9.0-11.0 1-5 drops 0.1% soln. in 90% alc. yellow blue
Thymolphthalein 9.4-10.6 1 drop 0.1% soln. in 90% alc. colorless blue
Nile blue 10.1-11.1 1 drop 0.1% aq. soln. blue red
Alizarin yellow 10.0-12.0 1 drop 0.1% aq. soln. yellow lilac
Salicyl yellow 10.0-12.0 1-5 drops 0.1% soln. in 90% alc. yellow orange-brown
Diazo violet 10.1-12.0 1 drop 0.1% aq. soln. yellow violet
Tropeolin O 11.0-13.0 1 drop 0.1% aq. soln. yellow orange-brown
Nitramine 11.0-13.0 1-2 drops 0.1% soln in 70% alc. colorless orange-brown
Poirrier's blue 11.0-13.0 1 drop 0.1% aq. soln. blue violet-pink
Trinitrobenzoic acid 12.0-13.4 1 drop 0.1% aq. soln. colorless orange-red
Primary References: Lange's Handbook of Chemistry, 8th Edition, Handbook Publishers Inc., 1952.
Volumetric Analysis, Kolthoff & Stenge, Interscience Publishers, Inc., New York, 1942 and 1947.
By: Princes Joy A. Nunez
Having trouble with acids and bases? You can’t determine if what category do they belong? Well, some of your questions can answer by this article.
What is an acid-base indicator? An acid-base indicator is a weak acid or a weak base. The undissociated form of the indicator is a different color than the iogenic form of the indicator. An Indicator does not change color from pure acid to pure alkaline at specific hydrogen ion concentration, but rather, color change occurs over a range of hydrogen ion concentrations. This range is termed the color change interval. It is expressed as a pH range.
How is an indicator used? Weak acids are titrated in the presence of indicators which change under slightly alkaline conditions. Weak bases should be titrated in the presence of indicators which change under slightly acidic conditions.
What are some common acid-base indicators? Several acid-base indicators are listed below, some more than once if they can be used over multiple pH ranges. Quantity of indicator in aqueous (aq.) or alcohol (alc.) solution is specified. Tried-and-true indicators include: thymol blue, tropeolin OO, methyl yellow, methyl orange, bromphenol blue, bromcresol green, methyl red, bromthymol blue, phenol red, neutral red, phenolphthalein, thymolphthalein, alizarin yellow, tropeolin O, nitramine, and trinitrobenzoic acid. Data in this table are for sodium salts of thymol blue, bromphenol blue, tetrabromphenol blue, bromcresol green, methyl red, bromthymol blue, phenol red, and cresol red.
Table of Common Acid-Base Indicators
Indicator pH Range Quantity per 10 ml Acid Base
Thymol Blue 1.2-2.8 1-2 drops 0.1% soln. in aq. red yellow
Pentamethoxy red 1.2-2.3 1 drop 0.1% soln. in 70% alc. red-violet colorless
Tropeolin OO 1.3-3.2 1 drop 1% aq. soln. red yellow
2,4-Dinitrophenol 2.4-4.0 1-2 drops 0.1% soln. in 50% alc. colorless yellow
Methyl yellow 2.9-4.0 1 drop 0.1% soln. in 90% alc. red yellow
Methyl orange 3.1-4.4 1 drop 0.1% aq. soln. red orange
Bromphenol blue 3.0-4.6 1 drop 0.1% aq. soln. yellow blue-violet
Tetrabromphenol blue 3.0-4.6 1 drop 0.1% aq. soln. yellow blue
Alizarin sodium
sulfonate 3.7-5.2 1 drop 0.1% aq. soln. yellow violet
α-Naphthyl red 3.7-5.0 1 drop 0.1% soln. in 70% alc. red yellow
p-Ethoxychrysoidine 3.5-5.5 1 drop 0.1% aq. soln. red yellow
Bromcresol green 4.0-5.6 1 drop 0.1% aq. soln. yellow blue
Methyl red 4.4-6.2 1 drop 0.1% aq. soln. red yellow
Bromcresol purple 5.2-6.8 1 drop 0.1% aq. soln. yellow purple
Chlorphenol red 5.4-6.8 1 drop 0.1% aq. soln. yellow red
Bromphenol blue 6.2-7.6 1 drop 0.1% aq. soln. yellow blue
p-Nitrophenol 5.0-7.0 1-5 drops 0.1% aq. soln. colorless yellow
Azolitmin 5.0-8.0 5 drops 0.5% aq. soln. red blue
Phenol red 6.4-8.0 1 drop 0.1% aq. soln. yellow red
Neutral red 6.8-8.0 1 drop 0.1% soln. in 70% alc. red yellow
Rosolic acid 6.8-8.0 1 drop 0.1% soln. in 90% alc. yellow red
Cresol red 7.2-8.8 1 drop 0.1% aq. soln. yellow red
α-Naphtholphthalein 7.3-8.7 1-5 drops 0.1% soln. in 70% alc. rose green
Tropeolin OOO 7.6-8.9 1 drop 0.1% aq. soln. yellow rose-red
Thymol blue 8.0-9.6 1-5 drops 0.1% aq. soln. yellow blue
Phenolphthalein 8.0-10.0 1-5 drops 0.1% soln. in 70% alc. colorless red
α-Naphtholbenzein 9.0-11.0 1-5 drops 0.1% soln. in 90% alc. yellow blue
Thymolphthalein 9.4-10.6 1 drop 0.1% soln. in 90% alc. colorless blue
Nile blue 10.1-11.1 1 drop 0.1% aq. soln. blue red
Alizarin yellow 10.0-12.0 1 drop 0.1% aq. soln. yellow lilac
Salicyl yellow 10.0-12.0 1-5 drops 0.1% soln. in 90% alc. yellow orange-brown
Diazo violet 10.1-12.0 1 drop 0.1% aq. soln. yellow violet
Tropeolin O 11.0-13.0 1 drop 0.1% aq. soln. yellow orange-brown
Nitramine 11.0-13.0 1-2 drops 0.1% soln in 70% alc. colorless orange-brown
Poirrier's blue 11.0-13.0 1 drop 0.1% aq. soln. blue violet-pink
Trinitrobenzoic acid 12.0-13.4 1 drop 0.1% aq. soln. colorless orange-red
Primary References: Lange's Handbook of Chemistry, 8th Edition, Handbook Publishers Inc., 1952.
Volumetric Analysis, Kolthoff & Stenge, Interscience Publishers, Inc., New York, 1942 and 1947.
By: Princes Joy A. Nunez
Formal Charges
In chemistry, a formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in a chemical bond are shared equally between atoms, regardless of relative electronegativity.
The formal charge of any atom in a molecule can be calculated by the following equation:
V = the number of valence electrons of the atom in ground state
N = the number of non-bonding valence electrons on this atom in the molecule B = the total number of electrons shared in covalent bonds with other atoms in the molecule. There are two electrons shared per single covalent bond.
Example Problem: In both cases the octet rule is satisfied for all of the atoms in the structure. Which is correct?
To decide, work out the formal charges on the carbon and oxygen in each molecule. Carbon has 4 valence electrons in the base atom, oxygen 6.
For the leftmost structure
* The carbon has four bonds, each worth 2 electrons, for a total of eight. It has no lone pairs. Thus, Cf = 4 - (0 + 1/2*8) =0
* The oxygen has two bonds, each worth 2 electrons, for a total of four. It has two lone pairs. Thus, Cf = 6 - (4 + 1/2*4) =0
For the rightmost structure
* The carbon has three bonds, each worth 2 electrons, for a total of six. It has one lone pair. Thus, Cf = 4 - (2 + 1/2*6) = -1
* The oxygen has three bonds, each worth 2 electrons, for a total of six. It has one lone pair. Thus, Cf = 6 - (2 + 1/2*6) = +1
The leftmost structure has the formal charges closer to zero, and thus is probably the correct structure.
By: Lorie Ann R. Anda
The formal charge of any atom in a molecule can be calculated by the following equation:
V = the number of valence electrons of the atom in ground state
N = the number of non-bonding valence electrons on this atom in the molecule B = the total number of electrons shared in covalent bonds with other atoms in the molecule. There are two electrons shared per single covalent bond.
Example Problem: In both cases the octet rule is satisfied for all of the atoms in the structure. Which is correct?
To decide, work out the formal charges on the carbon and oxygen in each molecule. Carbon has 4 valence electrons in the base atom, oxygen 6.
For the leftmost structure
* The carbon has four bonds, each worth 2 electrons, for a total of eight. It has no lone pairs. Thus, Cf = 4 - (0 + 1/2*8) =0
* The oxygen has two bonds, each worth 2 electrons, for a total of four. It has two lone pairs. Thus, Cf = 6 - (4 + 1/2*4) =0
For the rightmost structure
* The carbon has three bonds, each worth 2 electrons, for a total of six. It has one lone pair. Thus, Cf = 4 - (2 + 1/2*6) = -1
* The oxygen has three bonds, each worth 2 electrons, for a total of six. It has one lone pair. Thus, Cf = 6 - (2 + 1/2*6) = +1
The leftmost structure has the formal charges closer to zero, and thus is probably the correct structure.
By: Lorie Ann R. Anda

